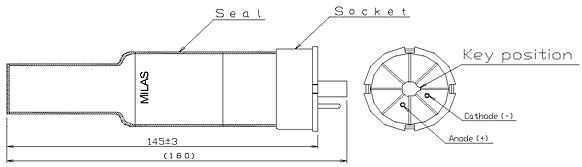
ġ3 The shape of the cathode which is hollow cylindrical concentrates the emitted radiation into a beam which passes through a quartz window all the way to the vaporized sample. Some sputtered atoms are in excited states and emit radiation characteristic of the metal as they fall back to the ground state. These gaseous ions bombard the cathode and eject metal atoms from the cathode in a process called sputtering. ġ0 Hollow Cathode Lamp cathode Anode Quartz window Pyrex body Anodeġ1 How it works Applying a potential difference between the anode and the cathode leads to the ionization of some gas atoms. Each element has its own unique lamp which must be used for that analysis. 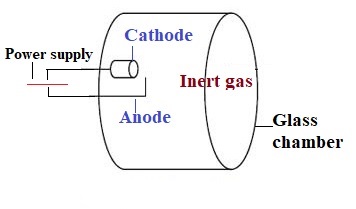
These are sealed in a glass tube filled with an inert gas (neon or argon ). It contains a tungsten anode and a hollow cylindrical cathode made of the element to be determined. ħ Schematic Diagram of an Atomic Absorption Spectrometerĭetector and readout device Light source (hollow cathode Lamp ) atomizer monochromatorĩ 1 – Light Source The light source is usually a hollow cathode lamp of the element that is being measured. Atomic absorption is a very common technique for detecting metals and metalloids in environmental samples.ĥ Elements detectable by atomic absorption are highlighted in pink in this periodic tableĪtomic absorption spectrometers have 4 principal components 1 - A light source ( usually a hollow cathode lamp ) 2 – An atom cell ( atomizer ) 3 - A monochromator 4 - A detector, and read out device. The analyte concentration is determined from the amount of absorption.Ĥ Concentration measurements are usually determined from a working curve after calibrating the instrument with standards of known concentration. The atoms absorb ultraviolet or visible light and make transitions to higher electronic energy levels. ģ - Atomic-absorption spectroscopy quantifies the absorption of ground state atoms in the gaseous state. It makes use of the absorption of light by these elements in order to measure their concentration. Atomic Absorption Spectroscopy Flame Atomic Emission Spectroscopy ICP Atomic Emission SpectroscopyĢ 1 BASIC PRINCIPLE ATOMIC ABSORPTION SPECTROSCOPY (AAS) is an analytical technique that measures the concentrations of elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
